  
Water weighs about 8.3 pounds per gallon. It is very dense.Īnother rather simple way to think of this is that if the density of water is 1 g/cc then the density of gold is 19.3 times greater than water. The old cowboy who robbed a stagecoach could not just throw a saddlebag of gold over his shoulder and ride off into the sunset. Thus a gallon of gold weighs 160.9 pounds. A pound contains 454 g of mass so 73501 g divided by 454 g/lb gives us the weight in pounds, 160.9 pounds. Since each cc has a mass of 19.3 g, a gallon of gold has a mass of 73051 g (3785 cc x 19.3 g/cc). If we want the density in English units we could use 11.1 oz/cubic inch as the density of gold.Ī gallon contains 3785 cubic centimeters or 231 cubic inches. For example, the mass density of gold is 19.3 g/cc, lead is 11.4 g/cc, copper is 9.0 g/cc, aluminum is 2.7 g/cc, water is 1.0 g/cc (1g/cc = 1 gram per cubic centimeter). The exact relationship is density = mass or weight divided by volume.ĭifferent materials have different densities. (Density tells us how much stuff is packed into how much space.) In the English system we use the weight of the material, while in the metric system the mass of the material is used. Density is a quantity that relates the amount of material present (mass or weight) to the amount of space (volume) it takes up. We can use either the English (weight) density or the metric (mass) density to answer the question. To answer this month’s question, we must refer to a quantity called density. Here on Earth the weight of an object is equal to the mass of the object times the acceleration due to Earth’s gravity, which is 9.8 m/s 2 or 32 ft/s 2. The two quantities mass and weight are directly related but not identical. People who use the metric system people usually speak of mass, while people using the English system of measurement usually speak of weight. 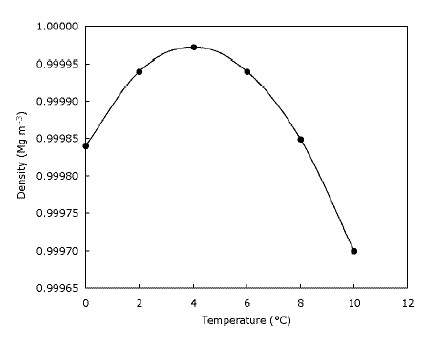
In everyday layman’s language people may use the word weight and mass interchangeably, however in science there is a distinct difference. Science is very specific and has specific definitions for its terminology. How much does a gallon of gold weigh? August 2000 Vocatio Center for Life Calling and Career.Office of Student Leadership & Engagement.In order to state how much something weighs, you have to say what it’s made from and where you’re weighing it.ĭoesn’t it make you wander how many gallon do ice weigh when melted? Here’s the converter. If you were to dissolve 10 teaspoons of sugar into a gallon of water, it would weigh a little more than 8.34 pounds – the weight of pure sugar is greater than pure salt. Water that has something else dissolved into it, like salt or sugar, will have a higher density and slightly different weight.įor instance, if you dissolve 10 teaspoons of salt into 1 gallon of water, then the resulting liquid will weigh about 8.37 pounds instead of 8.34 pounds. This, of course, assumes that we are talking about pure H2O (water molecules) – water with nothing else but hydrogen and oxygen atoms. And when the temperature is higher than 50 degrees, water weights heavier than at 50 degrees when the temperature is less than 50 degrees. For example, water in the summer season weighs more than when it’s cold. The weight of a liquid gallon of water in the UK and in the USA also varies in accordance with the room temperature and atmospheric pressure. You can see that when you buy a gallon, you’re getting a lot more water than when you buy a quart. On the other hand, if a gallon of water weighs a lot, how much does a quart of water weigh? One-quart weighs about 17.5 pounds. That’s a little more than a pound heavier than a gallon of milk. The metric equivalent is 3.785411784 liters. A US imperial gallon of water weighs approximately 8.34 pounds, a UK metric gallon of water weighs approximately 10.91 pounds.ġ Imperial gallon is equal to 4.54609 liters. This means that a gallon of water (16 cups) weighs about 128 fluid ounce and this will be about 11-to-12-pound weight (5-5.5 kilogram). Weight of a gallon of water is about 8.34 pounds (3.78 kg/L). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
